New research demonstrates role of anti-nephrin autoantibodies as causative factor and disease activity biomarker in minimal change disease and idiopathic nephrotic syndrome
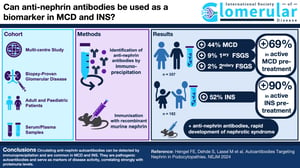
An international study using a novel assay technique reliably detected anti-nephrin autoantibodies in up to 90% of idiopathic nephrotic syndrome. This new research, presented May 25 at the 61st ERA Congress with simultaneous publication in the New England Journal of Medicine, was based on serum/plasma analysis of samples from a multi-center cohort of 357 adults with various glomerular diseases and 192 children with idiopathic nephrotic syndrome, plus 117 healthy controls. The study was led by a group from the University of Hamburg in a collaborative effort from ten other institutions across five countries, all connected via the International Society of Glomerular Disease.
Anti-nephrin autoantibodies are antibodies against the protein nephrin, which is integral to the structure and function of podocyte cells in the glomerulus. Using a novel hybrid technique of immunoprecipitation plus ELISA, the study detected anti-nephrin autoantibodies in approximately two-thirds of adult patients with minimal change disease (MCD) and active nephrotic syndrome before the initiation of immunosuppression, and in 90% of children with idiopathic nephrotic syndrome (INS) and active nephrotic syndrome before the initiation of immunosuppression. A smaller portion of patients with primary focal segmental glomerulosclerosis (FSGS) also exhibited anti-nephrin autoantibodies, while they were not detected in individuals with other glomerular conditions such as lupus nephritis or membranous nephropathy.
Furthermore, in anti-nephrin positive patients, anti-nephrin autoantibodies correlate with clinical disease activity and disease course during onset, remission and relapse of nephrotic syndrome. In a mouse model of active immunization with murine nephrin, animals developed anti-nephrin autoantibodies, followed by changes in nephrin phosphorylation, altered nephrin downstream signaling and the onset of a rapid nephrotic syndrome with a minimal change disease-like histological appearance. The investigators concluded from these findings that anti-nephrin autoantibodies are one of the permeability factors causing nephrotic syndrome in a large proportion of children and adults with idiopathic nephrotic syndrome or minimal change disease, as well as a smaller subset of individuals with FSGS. Notably, the mice became symptomatic at a relatively low level of autoantibodies after only one immunization, suggesting that auto-nephrin antibodies may mediate podocytopathies even at low concentrations and in the absence of traditional pathological antibody effects mediated by immune cells or the complement system.
Although autoantibodies targeting nephrin have been researched for over two decades, it has proven challenging to detect them reliably, fostering further discussion on their pathological relevance. The establishment of a reliable and sensitive assay for detection and quantification, its application to large cohorts of international origin and different glomerular diseases, the correlation to clinical disease activity and the creation of an animal model of anti-nephrin-mediated podocytopathy were therefore necessary to fully comprehend the role of anti-nephrin autoantibodies in glomerular diseases. In the future, anti-nephrin antibody measurement may enable nephrologists to make a noninvasive diagnosis, to guide the treatment of affected patients, and to help to reduce disease recurrence in patients after kidney transplantation. As study author Felicitas Hengel commented, it also finally enables affected individuals to “understand what they are suffering from, voiding the ‘idiopathic’ character of their disease.” Although the assay is not currently available outside of the study, the inventors plan to continue development with the ultimate goal of making it widely available.
These findings have promising implications for the future treatment of the related glomerular conditions, which currently have no disease-specific therapies and exact a hefty toll on affected individuals and families. The research team hopes that this improved pathobiological understanding of MCD and INS will enable clinicians to choose appropriate antibody-targeting therapies for patients and set the stage for the eventual development of therapeutics designed to specifically inhibit or counteract the autoantibodies in question.
Another promising avenue of exploration is the potential use of anti-nephrin antibodies as a disease activity marker, which could improve clinical care in several ways. First, measuring anti-nephrin antibodies during active nephrotic syndrome could help to diagnose anti-nephrin associated podocytopathy in patients who are ineligible for biopsy, for example due to young age, established anticoagulation medication or pregnancy. This is particularly important since INS and MCD often affect children and the loss of anticoagulation factors into the urine upon nephrotic syndrome can cause complications such as thrombosis, which in turn require the use of anticoagulants.
Second, the assessment of anti-nephrin as a prognostic marker in patients with INS, MCD, and FSGS will be of high interest. Prospective trials are necessary to determine if anti-nephrin antibodies can be used to predict a patient’s response to therapy, disease course, and risk of relapse. For patients with end-stage kidney disease who are being evaluated for a kidney transplant, the measurement of anti-nephrin autoantibodies could be a useful addition to the pre-transplant work-up to reduce the risk of post-transplant relapsing nephrotic syndrome. During follow-up of affected patients, the measurement of anti-nephrin autoantibodies could help to discriminate between a relapse of anti-nephrin-associated podocytopathy and other proteinuric disease in the transplanted kidney.
Lead authors Nicola Tomas and Tobias B. Huber emphasized that the nature of the project relied upon multi-institution collaboration from the beginning, as well as the work of previous researchers on anti-nephrin antibodies. Huber remarked that “research requiring larger cohorts in rare disease really needs organized professional networking structures, such the International Society of Glomerular Disease.” The research team looks forward to collaboration with additional investigators to conduct prospective studies aiming to bring these findings to the clinic and explore related questions such as the origin and triggers of anti-nephrin antibodies, the mechanism causing nephrin dysfunction upon antibody binding, and alternative etiologies in anti-nephrin negative MCD/INS patients.
Visual Abstract
FIND OUT MORE
For more information or to send a question to the study investigators, please visit our anti-nephrin antibodies FAQ page.
.png?width=300&name=ANAA%20banner%20(1920%20x%201080%20px).png)
COMMENTS